New Ozempic-Style Pill Outperformed Oral Semaglutide in Major Trial
We may have a new champion when it comes to pills that can manage both blood sugar levels and weight loss.
In a phase 3 clinical trial, once-daily orforglipron outperformed semaglutide tablets, the best-known oral version of the GLP-1 drugs.
GLP-1 (glucagon-like peptide-1) drugs are named after the natural hormone they mimic, helping curb appetite and stimulate insulin release to process sugar more efficiently.
As the active ingredient in treatments such as Wegovy and Ozempic, semaglutide has been hugely successful for tackling both diabetes and obesity. However, while it is available in tablet form, it’s most effective when taken via injection.
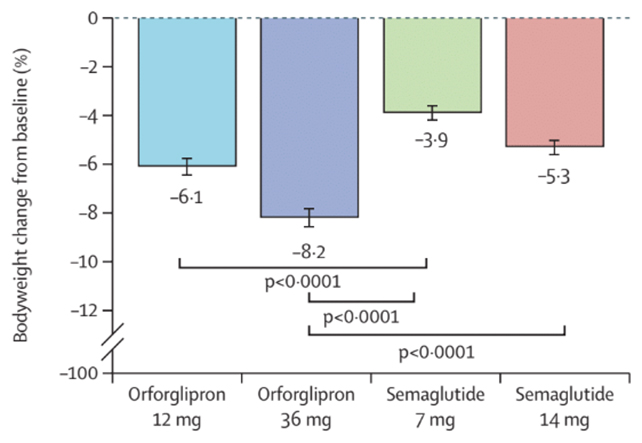
The new results, reported by an international team of researchers and funded by Eli Lilly, suggest orforglipron could become a leading oral alternative. Amongst 1,698 trial participants, people taking orforglipron saw bigger blood sugar improvements, and greater average weight loss (6–8 percent compared with 4–5 percent on semaglutide tablets).
“Together with previous research, the results of the ACHIEVE-3 study suggest that orforglipron represents an important advancement in the oral treatment landscape for type 2 diabetes,” write the researchers in their published paper.
The buzz has been growing around orforglipron: Another phase 3 trial carried out last year saw impressive results from the drug, though in that study it was compared to a placebo rather than semaglutide.
Phase 3 trials are considered a major step towards making drugs available to the public: They include larger groups of people than phase 1 and 2 trials, track benefits and risks, and compare drugs directly against other treatments.
Here, the participants were drawn from 131 different medical research centers and followed for a year. The trial was specifically aimed at people with type 2 diabetes, though there are plans to develop it as an obesity treatment too, in the future.
Orforglipron has another advantage over semaglutide: It doesn’t have to be taken on an empty stomach, making it more convenient for people it’s prescribed to.
The news from the trial wasn’t all positive for orforglipron, however. More people stopped taking orforglipron (9–10 percent) than stopped taking semaglutide (4–5 percent) during the trial, which was mostly down to side effects described as “gastrointestinal issues”.
“Although the safety profiles of both orforglipron and semaglutide were generally consistent with the GLP-1 receptor agonist class, the incidence of gastrointestinal events, discontinuations due to adverse events, and mean increase in pulse rate were higher with orforglipron than oral semaglutide,” write the researchers.
Regulatory approval will be required before orforglipron becomes available generally, but it’s getting close. The drug is also currently being evaluated as a way to improve cardiovascular health (something we’ve also seen with semaglutide).
There are now good reasons to believe that orforglipron can be even more effective than semaglutide in treating type 2 diabetes and obesity. The results add to growing evidence that GLP-1 drugs offer numerous other health benefits.
That said, the medications already on the market have also been linked to some major side effects: Wegovy, for example, has been connected to mental health issues and pancreatic damage in the past.
As always, it’s important that these medications, no matter how convenient and effective, are developed with patient safety as a priority. Trials like this are designed to map that trade-off more clearly, so doctors and regulators can judge where the benefits outweigh the risks.
Related: There’s a Surprising Link Between a Key Nutrient, Obesity, And Alzheimer’s Risk
“In conclusion, oral orforglipron was non-inferior and superior to semaglutide in terms of efficacy, with meaningful improvements in glycaemic control and weight reduction compared with oral semaglutide in patients with type 2 diabetes, and with larger improvements in cardiometabolic risk factors and simplified administration,” write the researchers.
The research has been published in The Lancet.
What did you think of this news? Leave a comment below and/or share it on your social media. This way, we can inform more people about the hottest things in technology, science, innovation, and gaming!
This news was originally published in:
Original source


