Forget Antibiotics: These Killer Cells Wipe Out Deadly Superbugs in a Day
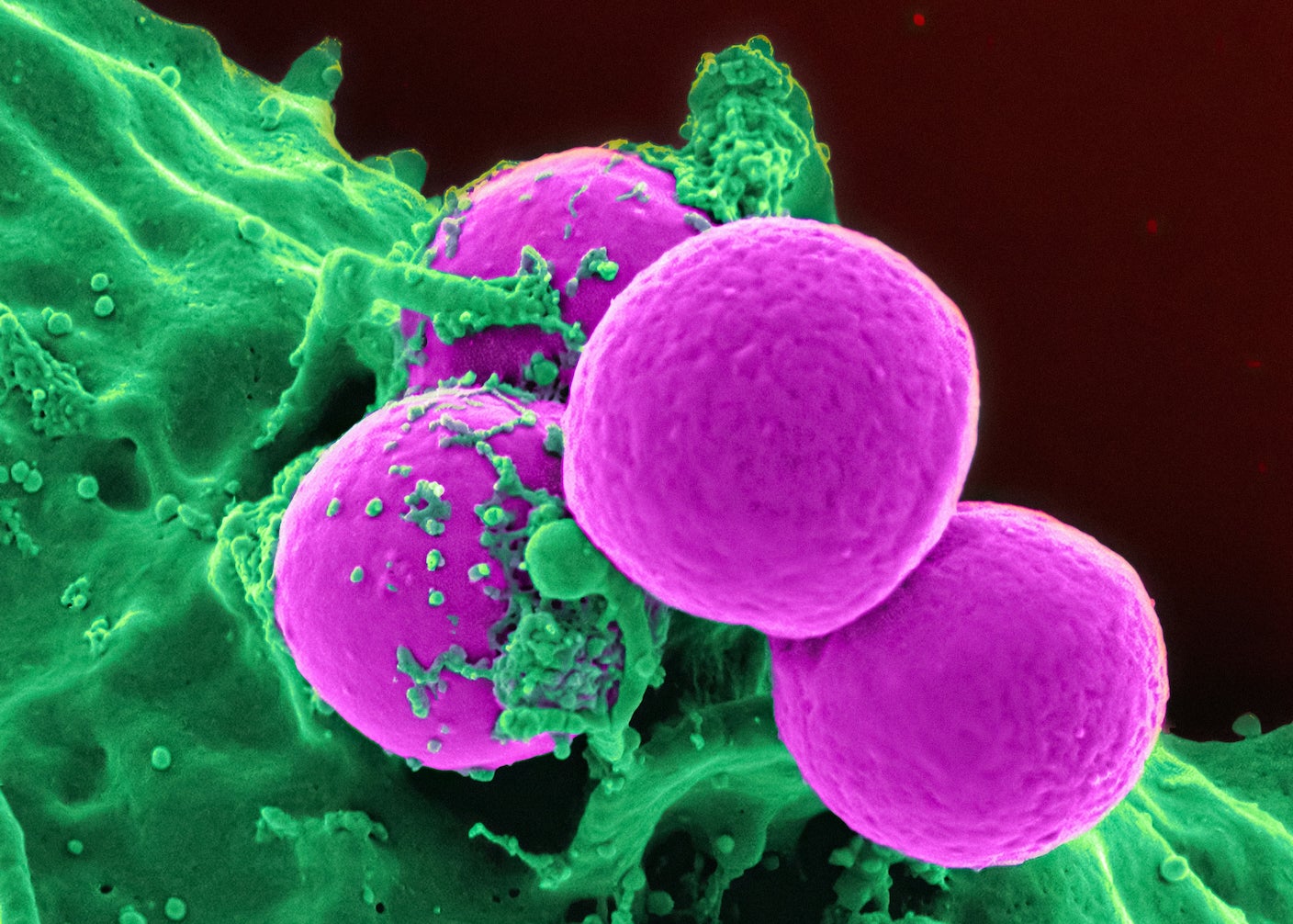
A mixture of bacteria lounge in a dish. Like the bugs populating our guts, most are benign or beneficial. But a deadly strain hides among them. These bacteria can easily escape last-line antibiotics, rapidly spread, and cause mayhem.
But in this case, a single dose of genetically engineered cells hunts them down and wipes out nearly the entire population in a day, while leaving all the other harmless cells alone.
This strategy, called minicell therapy, fights fire with fire: Researchers engineer hunter cells by stripping bacteria of the ability to replicate and then genetically loading them up with proteins to home in on dangerous foes. The cells grab their targets and inject toxins into them, releasing a hurricane of chemicals that causes the bacteria’s insides to collapse.
Developed by a team at the University of Oxford, the approach is completely different than current defenses against bacteria, making it harder for dangerous bugs to develop resistance. It’s also fairly simple to reprogram the engineered cells to target different bacterial strains.
The work shows how synthetic biology can bring wholly new weapons to the fight against deadly bacteria resistant to antibiotics, the authors wrote.
Brewing Crisis
Antimicrobial resistance is a critical global challenge projected to cause over 10 million deaths each year by 2050. Superbugs that dodge current treatments could spark the next pandemic, but our arsenal against them is dwindling.
Antibiotics work in different ways. Some puncture a bacteria’s protective wall, causing it to rupture. Others shut down protein production, damage DNA, or block metabolism to prevent growth.
Fighting bacteria is an evolutionary cat-and-mouse game. With time, bacterial genes mutate, and cells that escape one or many antibiotics grow, reproduce, and become dominant. Resistant bacteria can also share their genes with other cells to spread newly evolved defense systems.
Tweaking the chemical structure of an antibiotic buys some time. But what’s really needed are drugs that work in different ways. Unfortunately, the last new class of antibiotics now used in clinics dates back to the 1980s, followed by a decades-long lull. A novel class discovered in 2024 and the rise of AI-designed antibiotics have reinvigorated the field. But testing the candidates takes time, and they may not be able to catch up with the rapid spread of resistant bugs.
Other solutions are in the works. Phage therapy destroys bacteria with viruses and is already in clinical trials with initially positive results. Antibodies that neutralize bacterial toxins have also succeeded in early patient tests.
“However, these approaches face limitations such as stability issues, potential toxicity, and high manufacturing cost,” wrote the team.
A Smart Living Drug
Instead, they turned to an unusual creation called minicells to develop a completely new type of antibiotic. These cells, known more specifically as SimCells (short for “simple cells”), are made by stripping E. coli bacteria of their ability to replicate. Deleting an additional gene turns them into mini-SimCells that are roughly five times smaller.
Although some strains of E. coli can cause serious infections in the wild, the bacteria are reliable workhorses in research, synthetic biology, and biomanufacturing. They’re hardy, easy to grow, and plenty of tools already exist to genetically rewire their biology.
E. coli are also part of a growing effort to turn bacterial foes into living medicines to tackle conditions from metabolic disorders to cancer. Typically, benign probiotic strains are genetically modified to produce protein “bloodhounds” that help them seek out their cellular prey. Even familiar pathogens, like Salmonella, have been similarly repurposed. Once attenuated, they no longer cause disease and can be engineered to attack and inhibit cancer growth.
Though selected for safety, there’s a lingering risk of bacteria growing uncontrollably inside the body, triggering immune attacks, or escaping into the environment, wrote the team.
SimCells and their miniaturized cousin provide yet another layer of safety. Both are stripped of their native DNA so they can’t reproduce. But they retain all the other cellular machinery needed to survive and can make proteins from designer DNA. These cells are the perfect canvas for synthetic biology and have shown promise as shuttles for cancer drugs. One formulation even received “Fast-Track” status from the FDA to speed up development.
But they needed some biological rewiring to go after drug-resistant bacteria. The plan was to engineer SimCells and mini-SimCells that worked like “‘smart bioparticles’ to selectively eradicate pathogens, while sparing non-target bacteria,” the team wrote.
They first screened a library of nanobodies—tiny protein hooks that selectively latch onto a type of bacteria—and inserted genetic instructions for their chosen hooks into both types of designer cells. They then added another genetic payload encoding an enzyme that, with a small dose of aspirin, converted the drug into a chemical that produces hydrogen peroxide. After confirming the added genes, they introduced the cells into a dish full of bacteria.
The new cells were vicious. Their nanobodies guided them toward their prey and, when physically close, deployed their weapons. Nano-needles punctured the bacteria’s outer shell, releasing high doses of antimicrobial compounds—naturally made inside E. Coli as a defense system—into their foes. The cells also pumped out hydrogen peroxide for several days, forming a toxic environment that ruptured the bacteria and prevented stragglers from dividing.
This one-two punch slowed bacterial growth within six hours. After a day, 97 percent of the target bacteria were gone. Another day drove elimination to 99.9 percent.
“This antimicrobial strategy provides both immediate and sustained antimicrobial effects” that could prevent infections from coming back, wrote the team. In another test, the researchers engineered a range of SimCells and mini-SimCells dotted with different nanobodies that also reliably fought off multiple types of common drug-resistant bacteria.
But bacterial strains don’t exist in isolation. A kaleidoscope of beneficial bacteria support the gut, skin, and brain. These become collateral damage with classic antibiotic treatment. The new therapy was far more specific. Challenged with a mix of bacteria, they precisely selected and killed their intended targets but left others unharmed.
The therapy is still early. How the designer cells work inside the human body, especially alongside immune cells, remains to be tested. But thanks to a promising safety profile in a cancer clinical trial, the team is optimistic their infection-fighting versions are safe.
Though there weren’t any signs of resistance over the years-long study, the bacteria might eventually develop it. Researchers will have to track the cells over more time.
What did you think of this news? Leave a comment below and/or share it on your social media. This way, we can inform more people about the hottest things in technology, science, innovation, and gaming!
This news was originally published in:
Original source