Cannabis Compounds May Reverse Fatty Liver Disease, Study Suggests
The cannabis plant contains two powerful compounds that may reverse fatty liver disease in mice without causing any intoxication, according to new research.
The study, led by scientists at the Hebrew University of Jerusalem in Israel, found that both CBD (cannabidiol) and CBG (cannabigerol) can improve blood sugar control, reduce liver fat, and lower blood lipid levels in obese mice.
Strangely enough, both plant compounds achieved these results mostly independent of classical cannabinoid receptors, which are key regulators of communication between the gut and liver.
Instead, daily injections of either CBD or CBG into the abdomen of mice enhanced the production of phosphocreatine, a form of creatine released by the liver to help replenish energy supplies and maintain cellular health.
After mice were fed a high-fat diet, CBD and CBG restored some of their liver function after four weeks.
CBG proved particularly effective, reducing body fat, lowering ‘bad’ cholesterol, and increasing insulin sensitivity in obese mice significantly more than CBD.
“Our findings identify a new mechanism by which CBD and CBG enhance hepatic [liver] energy and lysosomal function,” says pharmacist and senior author Joseph Tam.
“This dual metabolic remodeling contributes to improved liver lipid handling and highlights these compounds as promising therapeutic agents for metabolic dysfunction-associated steatotic liver disease (MASLD).”
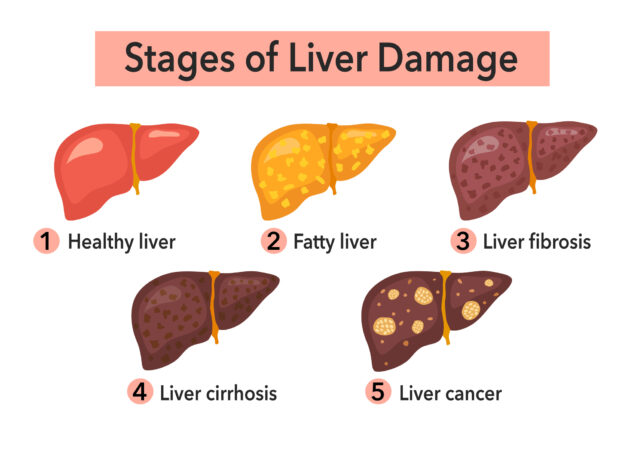
MASLD occurs when fat builds up in the liver. It is distinct from alcohol-related liver disease, and it has become the most common chronic liver disorder in the world, impacting roughly a third of the global adult population.
But MASLD is not only a liver condition; it is also a systemic metabolic disorder, and in recent years, animal studies have suggested that natural, bioactive compounds from the cannabis plant may hold promise as treatments.
CBD is one of the best-known and most studied compounds from the cannabis plant, and while research is still limited and conflicted, some studies suggest this compound can have beneficial metabolic effects.
CBG, meanwhile, has only recently emerged as an alternative cannabis compound with the potential to improve health outcomes even more than CBD. It is sometimes dubbed the “mother of all cannabinoids” because it rapidly metabolizes into CBD and the psychoactive compound in cannabis, THC.
Neither CBD nor CBG seems to be active in the central nervous system (at least not in their purest form), which means on their own they don’t trigger a ‘high’ in human patients like THC does. This is another benefit to their potential use as medicine.
“This study is the first to demonstrate that phytocannabinoids can reprogram hepatic energy buffering,” argue the authors of the study.
In previous research on rodents, creatine supplementation showed some ability to resolve MASLD, but it had the opposite outcome for fatty liver disease triggered by alcohol use.
The current study in MASLD-like mice supports these results, finding that some cannabis compounds can protect the liver by shifting energy toward the synthesis of phosphocreatine and restoring cellular mechanisms that clear fats from the organ.
Whether those results hold up in humans remains to be seen. Today, CBD products on the market are not very closely regulated, and some may not be sold in their purest form.
What’s more, these products are often oral droplets, and it’s unclear whether the medicine would have the same impact if swallowed as opposed to injected straight into the abdomen.
Related: ‘Mother of All Cannabinoids’ Tested in First Human Clinical Trial
Perhaps if further research can reveal how CBD and CBG have these impacts on liver function, a novel drug can be created to mimic their effects in a way that is easily delivered and safe for use.
“Despite the increasing clinical burden of MASLD, no pharmacological treatments have been approved to date,” the study authors write.
“This therapeutic gap underscores the urgent need for novel pharmacological agents that can target the underlying mechanisms of disease progression.”
The study was published in the British Journal of Pharmacology.
What did you think of this news? Leave a comment below and/or share it on your social media. This way, we can inform more people about the hottest things in technology, science, innovation, and gaming!
This news was originally published in:
Original source