These Genetically Engineered Brain Cells Devour Toxic Alzheimer’s Plaques
Alzheimer’s disease and cancer have something in common: They’re hard to treat.
Despite decades of research, both still plague humanity, robbing people of longer, healthier lives. In Alzheimer’s, a protein called amyloid forms toxic clumps in the brain. Eventually, neurons supporting memory, decision-making, and movement wither.
Whether amyloid clumps cause Alzheimer’s is still hotly debated. Drugs that clear the proteins have slowed disease progress, but only mildly. The FDA recently approved two such drugs for early stage patients. The approvals were controversial, however, due to risks, like brain bleeds.
As scientists have struggled with Alzheimer’s, blood cancer treatments have undergone a revolution thanks to a therapy called CAR T. The treatment genetically engineers a patient’s T cells to hunt and destroy a handful of previously untreatable blood cancers.
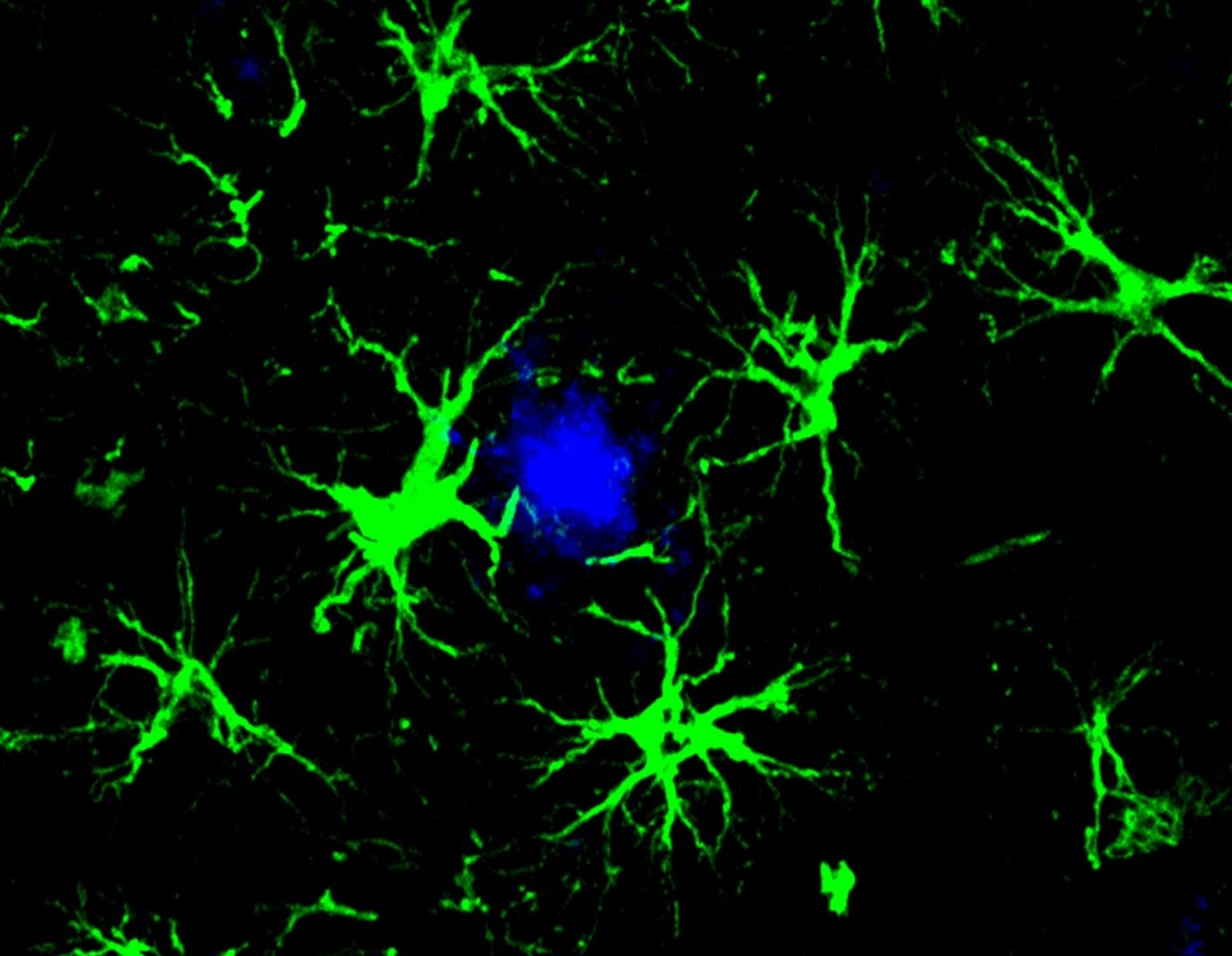
Taking a page from CAR T, a team at the Washington University School of Medicine, St. Louis has now transformed brain cells, known as astrocytes, into amyloid-gobbling machines.
A single injection into mice with Alzheimer’s prevented the formation of amyloid clumps in the brain during the disease’s early stages. In mice whose brains were already riddled with the toxic plaques, the treatment cut amyloid levels roughly in half.
“This study marks the first successful attempt at engineering astrocytes to specifically target and remove amyloid beta plaques in the brains of mice with Alzheimer’s disease,” study author Marco Colonna said in a press release.
Mysterious Origins
Alzheimer’s has baffled scientists for over a century. Genetics play a role. Some versions of a gene called APOE protect the brain against the disease; others exacerbate it. Inflammation in the skin, lungs, or gut may be an early trigger. Damage to waste-cleaning cells that normally wash toxic proteins out of the brain could also contribute to symptoms.
But the reigning theory of what causes Alzheimer’s is amyloid buildup. These sticky proteins aren’t inherently evil. At low levels, they tweak how neurons connect to make memories, support brain healing after injury, and may ward off infections.
In Alzheimer’s, however, amyloid clumps into toxic waste. The brain’s immune cells can initially clear them up. But amyloid eventually overwhelms these cells and causes them to spew inflammatory molecules that exacerbate the disease.
Amyloid clumps ignite a molecular cascade, leading to brain inflammation and ultimately neuronal loss, wrote Jake Boles and David Gate at Northwestern University, who were not involved in the study.
Existing drugs target amyloid proteins with antibodies that either physically prevent amyloid from clumping or mark the proteins for destruction by the immune system. But patients need regular treatment, and the risk of brain bleeds or stroke-like symptoms cause many to opt out.
Engineering brain cells to do the job could be a lasting solution.
In CAR T, scientists genetically engineer T cells to produce special proteins, or CARs, that latch onto cancer cells and destroy them. These have two sections: One outside the cell to recognize targets, and another inside the cell to trigger a biological effect. For CAR T cells, the interior trigger releases a blast of molecular bullets that destroys cancers or pathogens—and in one study, even amyloid proteins in mice. Another recent study modified a different type of immune cell to clear the toxic clumps, although its safety is still unknown.
These test cases suggest that CAR technology could reduce toxic amyloid buildup. But they used cell types that weren’t native to the brain and needed additional chemicals to keep them working. This mismatch could make treatment more complex and risk side effects.
In contrast, “genetically engineering resident brain cells to target…[amyloid]…could circumvent these persistence challenges,” wrote Boles and Gate.
Meet CAR-A
The team turned to astrocytes. These support cells help repair brain injuries, provide nutrients to neurons, and impact memory and cognition. They also eat up dead cell fragments and some proteins, though they’re less efficient at this task than the brain’s immune cells.
To boost astrocytes’ appetite for amyloid, the team tested multiple CAR designs. Genetic instructions for two of these were then packaged into a harmless virus for delivery and injected into the veins of mice modeling Alzheimer’s disease.
Inside the brain, the treatment transformed naturally occurring astrocytes into CAR-A cells. In mice two and a half months old, roughly late adolescence in human years, the cells prevented amyloid clumps for at least three months. And a single shot slashed amyloid levels in half for older mice already suffering buildup. The treatment also protected neurons from further damage.
Though tailored to astrocytes, the gene therapy also caused the brain’s immune cells to more readily devour amyloid plaques, swept away malfunctioning ones, and lowered inflammation. Shifting some of the amyloid-clearing burden from immune cells to astrocytes could create a less toxic environment in the Alzheimer’s brain, wrote Boles and Gate.
Anti-amyloid antibodies in current Alheimer’s drugs struggle to tunnel through the blood-brain barrier. But CAR-A cells are made inside the brain with minimal blood vessel damage, which could also lower the chances of deadly side effects.
“Consistent with the antibody drug treatments, this new CAR-astrocyte immunotherapy is more effective when given in the earlier stages of the disease,” said study author David Holtzman. “But where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins in mice.”
Long Road Ahead
While the new approach may improve on current treatments, it ran into the same problem: The shot reduced amyloid clumps without significantly improving memory or mood.
The results mirror those seen in anti-amyloid drug trials. It could be because over-zealous CAR-A cells nibble on neurons too and destroy their connections, offsetting any benefit. Or more fundamentally, it could be that targeting amyloid alone isn’t enough. Tau proteins, for example, also aggregate in neurons as the disease goes on, and higher levels are tied to mental decline.
Early treatments targeting tau proteins have universally failed. But the CAR-A platform could be redesigned to go after tau in a combination therapy to wipe out both toxic proteins.
Beyond Alzheimer’s, a similar strategy may be work in other brain diseases to kill brain cancers. The team is now fine-tuning their designs to better detect targets.
“As CAR technologies mature and the ability to selectively neutralize toxic proteins improves, these approaches hold substantial promise for AD [Alzheimer’s disease] and other neurodegenerative disorders,” wrote Boles and Gate.
What did you think of this news? Leave a comment below and/or share it on your social media. This way, we can inform more people about the hottest things in technology, science, innovation, and gaming!
This news was originally published in:
Original source