Popular Anti-Aging Supplement May Fuel Cancer Growth – Here’s Why
A group of natural compounds attracting attention for their anti-aging potential has a dark side.
New research shows how a family of chemicals called polyamines speeds up the growth of cancer cells. Led by a team from the Tokyo University of Science in Japan, the study offers some significant new insights for both cancer treatment research and the development of anti-aging therapies.
Polyamines are essential molecules found in all living cells. Including compounds with colorful names like spermidine and putrescine, they regulate processes involving cell growth and protein synthesis.
Past studies using animal models have linked spermidine to improved lifespans, better health, and reduced age-related memory loss. You can also find it in numerous over-the-counter supplements promoting health and well-being.
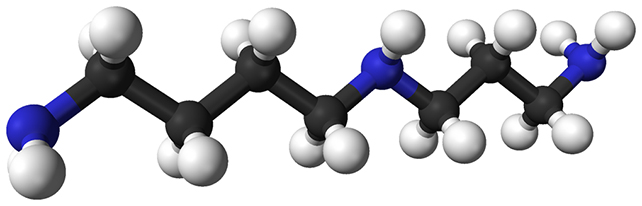
However, researchers have also linked spermidine and the other polyamines to cancer spread. The goal of this new study was to take a closer look at that relationship and the specific biological changes that might be occurring – especially how cancer cells burn through sugar without using oxygen, even when sufficient oxygen is present.
“Changes in polyamine metabolism are correlated with various pathologies, including cancer and age-related conditions,” write the researchers in their published paper.
“However, how polyamines directly modulate genes that affect cancer progression, in particular aerobic glycolysis-dependent cell growth, remains unclear.”
The researchers used lab cultures of cervical cancer and breast cancer cells to investigate how cancerous tissues responded to various tweaks and modifications, including the knock-on effects on a pair of proteins called eIF5A1 and eIF5A2.
While these two proteins are fundamentally very similar, sharing almost identical amino acid sequences, eIF5A1 has been identified as being crucial to healthy cells, while eIF5A2 is thought to be important to cancer growth and development.
As polyamines affect the production of these two proteins, the researchers suspected the proteins might be behind spermidine’s dual role as both an anti-aging compound and a cancer driver.
Using drugs and genetic editing, the levels of polyamines and these two proteins were altered in the lab cancer cells.
The cell behavior showed that polyamines push cancer cells towards an aerobic glycolysis mode (cancer’s favorite way of fueling growth), and boost levels of the eIF5A2 protein – primarily by removing a natural ‘brake’ on its production in the form of an RNA molecule called miR-6514-5p.
Removing polyamines or eIF5A2 from the cells reduced the growth of cancer cells significantly, while returning spermidine restored the cancer’s expansion. That’s strong evidence that spermidine may have a critical role in tumor proliferation.
“The biological activity of polyamines via eIF5A differs between normal and cancer tissues,” says biochemist Kyohei Higashi, from Tokyo University of Science.
“In normal tissues, eIF5A1, activated by polyamines, activates mitochondria via autophagy, whereas in cancer tissues, eIF5A2, whose synthesis is promoted by polyamines, controls gene expression at the translational level to facilitate the proliferation of cancer cells.”
This research stops a long way short of saying spermidine and polyamines in general cause cancer. Rather, it’s showing that when cancer has already started through some biological malfunction, the cancer cells can hijack the advantages of polyamines to survive and spread further.
The extra details of the roles of eIF5A2 and miR-6514-5p give researchers the opportunity to test out new treatment targets. It’s possible that with some drug-induced changes to these proteins and molecules, the mechanisms that cancer relies on could be disrupted.
Related: Two Cancers Have Links With Talc. An Expert Explains The Science.
Any time that potential new treatment targets like these are identified, our chances of developing effective cancer drugs go up. A great deal of careful research is needed along the way, though, to make sure healthy cells aren’t adversely affected – and this research is limited to cell cultures in the lab, for now.
“Our findings reveal an important role for eIF5A2, regulated by polyamines and miR-6514-5p, in cancer cell proliferation, suggesting that the interaction between eIF5A2 and ribosomes, which regulate cancer progression, is a selective target for cancer treatment,” write the researchers.
The research has been published in the Journal of Biological Chemistry.
What did you think of this news? Leave a comment below and/or share it on your social media. This way, we can inform more people about the hottest things in technology, science, innovation, and gaming!
This news was originally published in:
Original source